|
2/27/2023 0 Comments Amine functional group 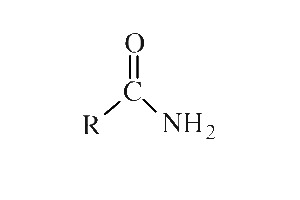
Functional groups are extremely useful in determining the behavior and physical properties of a given molecule. | Key Concepts and Summary | Glossary | End of Section Exercises |Ī functional group is a specific set of atoms that has characteristic properties. Categorize primary, secondary, and tertiary alcohols and amines.| Summary Table | Intermolecular Forces | Using suffix information, associate structures containing a single functional group to their names.| Alcohol | Ether | Aldehyde and Ketone | Carboxylic Acid and Ester | Amine | Amide | Identify functional groups in molecular structures and draw organic structures containing selected functional groups.For example, the COOR is ester it can not be recognized as a “ketone” plus an “ether”. For these three functional groups, it is important to remember that the “W” part has to be considered together with the C=O, since overall they define the functional group. The other three: ester, anhydride and amide, are all derivatives of carboxylic acid, meaning they can be prepared with carboxylic acid as the starting material. When an OH group is connected with C=O, the whole COOH is called a carboxylic acid functional group. The last four functional groups are related in terms of structures and chemical properties. Figure 2.3j Ketone and cyclic ketone examples Ketone can also be in a cyclic structure. Ketone, on the other hand, must be in the middle position to ensure both sides of the C=O groups are connected with R groups. Because H has to be connected on one side of the C=O group in aldehyde, aldehyde can only be at the end of a structure. Aldehyde can be regarded as a special case of ketone since “H” can be regarded as an R with zero carbon. Figure 2.3i General structure of functional groups containing c=o bondĪldehyde and ketone are similar in terms of their structures and properties. It is usually more challenging to identify and draw these functional groups correctly because they are similar. Figure 2.3h Primary, secondary, & tertiary amineįor the functional groups in the 2 nd part of Table 2.2, they all have a common structural unit of a carbonyl group C=O the different structure of “W” in the general formula determines the nature of the functional group. 
Amines can also be referred to with common names. Amine can be primary (1°), secondary (2°) or tertiary (3°) depending on how many R groups are connected with nitrogen. When the hydrogen atom(s) in NH 3 is replaced with R groups, it produces amine. Nitrile has a C≡N triple bond, and therefore can only be at the end of a structure, while nitro (NO 2) can be in any position on the carbon chain or ring.Īmine is the organic derivative of ammonia, NH 3. Figure 2.3g Cyclic ether examplesīoth nitrile and nitro groups contain nitrogen atoms, and they can be easily confused. It may not be that intuitive to recognize the following structure as ether, and labelling the carbon atom will be helpful for identification. Figure 2.3f diethyl etherĮther can be in a cyclic structure as well. When the two alkyl groups are the same, they can be combined as “dialkyl”. Compounds with ether as the only functional group are usually referred to with the common name “alkyl alkyl ether”. In ether, the O atom connects with two carbon-containing R groups through two C-O σ bonds. Figure 2.3e 1° alcoholĪnother functional group that contains the oxygen atom in single bonds is ether. Depending on the position of the OH group, alcohols can also be categorized as primary (1°), secondary (2°) or tertiary (3°). In organic chemistry, the term alcohol refers to a compound containing the OH (hydroxy) group. Alcohol is a functional group that you are probably familiar with. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
